Medical News
On November 14, 2019, the Food and Drug Administration granted accelerated approval to zanubrutinib (BRUKINSA, BeiGene, Ltd.) for adult patients with mantle cell lymphoma (MCL) who have received at least one prior therapy. Efficacy was evaluated in BGB-3111-206 (NCT03206970), a phase 2 open-label, multicenter, single-arm trial of 86 patients with MCL who received at least
Read More
On November 8, 2019, the Food and Drug Administration approved luspatercept-aamt (REBLOZYL, Celgene Corp.) for treatment of anemia in adult patients with beta thalassemia who require regular red blood cell transfusions. Efficacy was evaluated in the BELIEVE trial (NCT02604433), a multicenter, randomized, double-blind, placebo-controlled trial enrolling 336 adult patients with beta thalassemia requiring regular red
Read More
On August 16, 2018, the Food and Drug Administration granted accelerated approval to nivolumab (Opdivo, Bristol-Myers Squibb Company Inc.) for patients with metastatic small cell lung cancer (SCLC) with progression after platinum-based chemotherapy and at least one other line of therapy. Approval was based on demonstration of a durable overall response rate (ORR) in a
Read More
On July 10, 2018, the Food and Drug Administration granted accelerated approval to ipilimumab (YERVOY, Bristol-Myers Squibb Company Inc.) for use in combination with nivolumab for the treatment of patients 12 years of age and older with microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) metastatic colorectal cancer (mCRC) that has progressed following treatment with
Read More
On November 21, 2018, the Food and Drug Administration granted accelerated approval to venetoclax (VENCLEXTA, AbbVie Inc. and Genentech Inc.) in combination with azacitidine or decitabine or low-dose cytarabine for the treatment of newly-diagnosed acute myeloid leukemia (AML) in adults who are age 75 years or older, or who have comorbidities that preclude use of
Read More
On April 11, 2019, the Food and Drug Administration approved pembrolizumab (KEYTRUDA, Merck Inc.) for the first-line treatment of patients with stage III non-small cell lung cancer (NSCLC) who are not candidates for surgical resection or definitive chemoradiation or metastatic NSCLC. Patients’ tumors must have no EGFR or ALK genomic aberrations and express PD-L1 (Tumor
Read More
On March 18, 2019, the Food and Drug Administration approved atezolizumab (TECENTRIQ, Genentech Inc.) in combination with carboplatin and etoposide, for the first-line treatment of adult patients with extensive-stage small cell lung cancer (ES-SCLC). Approval was based on IMpower133 (NCT02763579), a randomized (1:1), multicenter, double-blind, placebo-controlled trial in 403 patients with ES-SCLC who received no
Read More
On May 2, 2019, the Food and Drug Administration approved ivosidenib (TIBSOVO, Agios Pharmaceuticals, Inc.) for newly-diagnosed acute myeloid leukemia (AML) with a susceptible IDH1 mutation, as detected by an FDA-approved test, in patients who are at least 75 years old or who have comorbidities that preclude the use of intensive induction chemotherapy. Approval was
Read More
On March 18, 2019, the Food and Drug Administration approved atezolizumab (TECENTRIQ, Genentech Inc.) in combination with carboplatin and etoposide, for the first-line treatment of adult patients with extensive-stage small cell lung cancer (ES-SCLC). Approval was based on IMpower133 (NCT02763579), a randomized (1:1), multicenter, double-blind, placebo-controlled trial in 403 patients with ES-SCLC who received no
Read More
On December 6, 2018, the Food and Drug Administration approved atezolizumab (TECENTRIQ, Genentech, Inc.), in combination with bevacizumab, paclitaxel, and carboplatin for the first-line treatment of patients with metastatic non-squamous, non-small cell lung cancer (NSq NSCLC) with no EGFR or ALK genomic tumor aberrations. Approval was based on the IMpower150 trial (NCT02366143), an open-label, randomized
Read More
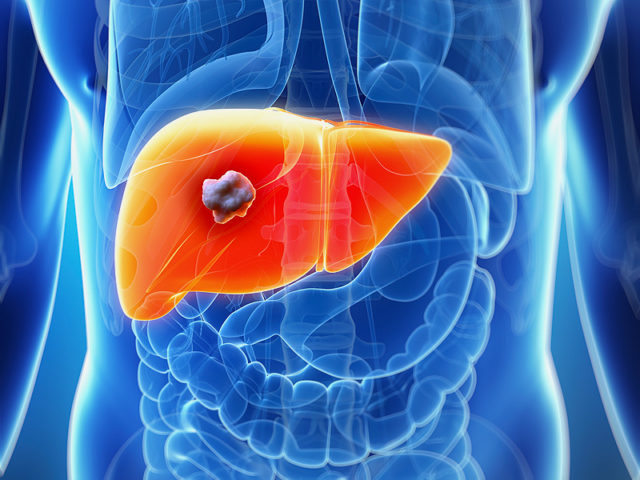
On January 14, 2019, the Food and Drug Administration approved cabozantinib (CABOMETYX, Exelixis, Inc.) for patients with hepatocellular carcinoma (HCC) who have been previously treated with sorafenib. Approval was based on CELESTIAL (NCT01908426), a randomized (2:1), double-blind, placebo-controlled, multicenter trial in patients with HCC who had previously received sorafenib and had Child Pugh Class A
Read More
The U.S. Food and Drug Administration today approved Gamifant (emapalumab-lzsg) for the treatment of pediatric (newborn and above) and adult patients with primary hemophagocytic lymphohistiocytosis (HLH) who have refractory, recurrent or progressive disease or intolerance with conventional HLH therapy. This FDA approval is the first for a drug specifically for HLH. “Primary HLH is a
Read More
On August 16, 2018, the Food and Drug Administration approved lenvatinib capsules (Lenvima, Eisai Inc.) for first-line treatment of patients with unresectable hepatocellular carcinoma (HCC). Approval was based on an international, multicenter, randomized, open-label, non-inferiority trial (REFLECT; NCT01761266) conducted in 954 patients with previously untreated, metastatic or unresectable HCC. Patients were randomized (1:1) to receive
Read More
Today, nivolumab (Opdivo) received approval from the U.S. Food and Drug Administration (FDA) for patients with metastatic small cell lung cancer (SCLC) whose cancer has progressed after platinum-based chemotherapy and at least one other line of therapy. Approval for this indication has been granted under accelerated approval based on overall response rate and duration of response. This approval
Read More
The U.S. Food and Drug Administration today approved Tibsovo (ivosidenib) tablets for the treatment of adult patients with relapsed or refractory acute myeloid leukemia (AML) who have a specific genetic mutation. This is the first drug in its class (IDH1 inhibitors) and is approved for use with an FDA-approved companion diagnostic used to detect specific
Read More
On June 8, 2018, the Food and Drug Administration granted regular approval to venetoclax (VENCLEXTA, AbbVie Inc. and Genentech Inc.) for patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL), with or without 17p deletion, who have received at least one prior therapy. Approval was based on MURANO (NCT02005471), a randomized (1:1), multicenter,
Read More
– Breakthrough Product is a Major Advance in the Treatment of Patients Hospitalized with Life-Threatening Bleeding – Rivaroxaban Apixaban – Company to Host Conference Call on Friday, May 4, 2018 at 8:30 a.m. ET – SOUTH SAN FRANCISCO, Calif., May 03, 2018 (GLOBE NEWSWIRE) — Portola Pharmaceuticals, Inc.® (Nasdaq:PTLA) today announced that the U.S. Food and Drug Administration
Read More
The U.S. Food and Drug Administration today approved Imfinzi (durvalumab) for the treatment of patients with stage III non-small cell lung cancer (NSCLC) whose tumors are not able to be surgically removed (unresectable) and whose cancer has not progressed after treatment with chemotherapy and radiation (chemoradiation). “This is the first treatment approved for stage III
Read More
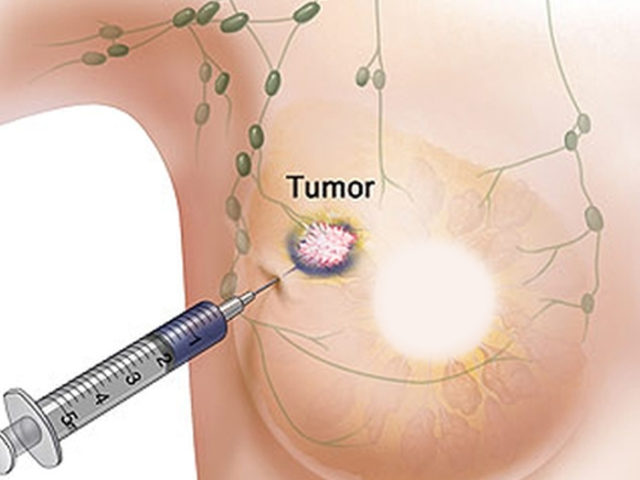
The U.S. Food and Drug Administration today approved Verzenio (abemaciclib) to treat adult patients who have hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced or metastatic breast cancer that has progressed after taking therapy that alters a patient’s hormones (endocrine therapy). Verzenio is approved to be given in combination with an endocrine
Read More
On Aug. 17, 2017, the U.S. Food and Drug Administration granted regular approval to olaparib tablets (Lynparza, AstraZeneca) for the maintenance treatment of adult patients with recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer, who are in a complete or partial response to platinum-based chemotherapy. With the addition of the new indication, a tablet
Read More
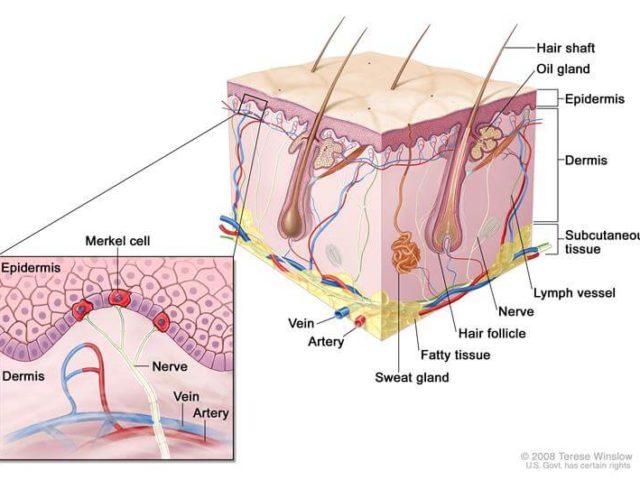
The Food and Drug Administration (FDA) has approved the immunotherapy drug avelumab (Bavencio®) for the treatment of some patients with a rare and aggressive skin cancer known as Merkel cell carcinoma. It is the first FDA-approved treatment for this disease. The approval was based on results from a clinical trial that included 88 patients with metastaticMerkel cell carcinoma who had previously been treated
Read More
The U.S. Food and Drug Administration today approved Rydapt (midostaurin) for the treatment of adult patients with newly diagnosed acute myeloid leukemia (AML) who have a specific genetic mutation called FLT3, in combination with chemotherapy. The drug is approved for use with a companion diagnostic, the LeukoStrat CDx FLT3 Mutation Assay, which is used to
Read More
The U.S. Food and Drug Administration today expanded the approved use of Stivarga (regorafinib) to include treatment of patients with hepatocellular carcinoma (HCC or liver cancer) who have been previously treated with the drug sorafenib. This is the first FDA-approved treatment for a liver cancer in almost a decade. “Limited treatment options are available for
Read More
The U.S. Food and Drug Administration today approved Nerlynx (neratinib) for the extended adjuvant treatment of early-stage, HER2-positive breast cancer. For patients with this type of cancer, Nerlynx is the first extended adjuvant therapy, a form of therapy that is taken after an initial treatment to further lower the risk of the cancer coming back.
Read More